 Its units are the change in energy ( \Delta E \text. This random motion, this entropy, is represented by S, and is a measure of the energy that is unable to do work. This law explains that concentrated energy has a tendency to become dispersed energy in the form of random thermal motion.ģ. 
This means that the entropy of a closed or isolated system will always increase over time. The second law states that the entropy of the universe is always increasing. It states that energy cannot be created or destroyed, only transferred between types of energy, such as heat and work.Ģ. The first law of thermodynamics is the theory of conservation of energy. Finally, we will revisit the topic of Gibbs free energy, of which you should already have a decent understanding, and how it relates to enthalpy vs. If the process takes place over a range of temperature, the quantity can be evaluated by adding bits of entropies at various temperatures. Thus, entropy has the units of energy unit per Kelvin, J K -1. 
Once you have a firm hold on the definition of enthalpy, we will discuss entropy and look at entropy practice questions. Entropy is the amount of energy transferred divided by the temperature at which the process takes place. Entropy is a factor of state function i.e. A system with a great degree of disorderliness has more entropy. Then we will talk about the definition of enthalpy and how to calculate it using enthalpy practice questions. Entropy Change can be defined as the change in the state of disorder of a thermodynamic system that is associated with the conversion of heat or enthalpy into work. This tutorial explains the theory behind enthalpy and entropy using the laws of thermodynamics. The change in entropy describes whether the system is heading towards greater disorder or less disorder, if it is positive, it heads towards more disorder but. The change of entropy of a system during a process is calculated using the equation: ΔS = S_final - S_initial, where ΔS is the change in entropy, S_final is the entropy of the system after the process is complete, and S_initial is the entropy of the system before the process started.When you get to the thermodynamics section of AP® Chemistry, one of the hardest things to remember is the definition of enthalpy vs. However, a reaction that produces fewer molecules from more molecules will decrease the entropy of the system. 
Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic quantities. As with standard heat capacities, we measure entropy amounts as energy at a given temperature for a given amount of the substance, in this case, moles. For example, a reaction that produces more molecules from fewer molecules will typically increase the entropy of the system because there are more possibilities for arranging the molecules. Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system. The second law of thermodynamics states that the total entropy of a closed system always increases over time, which means that the system becomes more disordered or randomized over time.Īlso, chemical reactions can increase or decrease the entropy of a system. The enthalpy and entropy change for the reaction are 30 KJ/mol and 105 J/K/mol, find out if T 285.7K. So, entropy is a measure of the number of possible arrangements of a system that are consistent with its overall properties, such as temperature, pressure, and volume. The Enthalpy and entropy changes of a reaction are 40.63 KJmol1 and 108.8JK1mol1, the value of G is positive and hence the reaction is nonspontaneous. 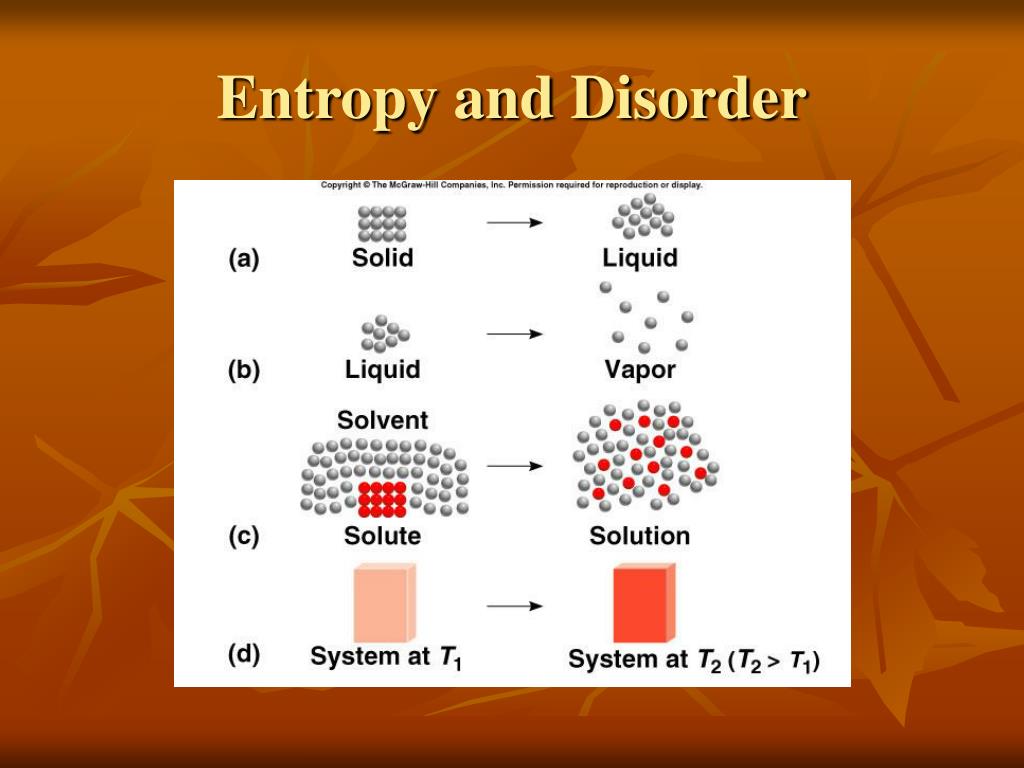
Entropy in simple terms is the disorder or randomness in a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
